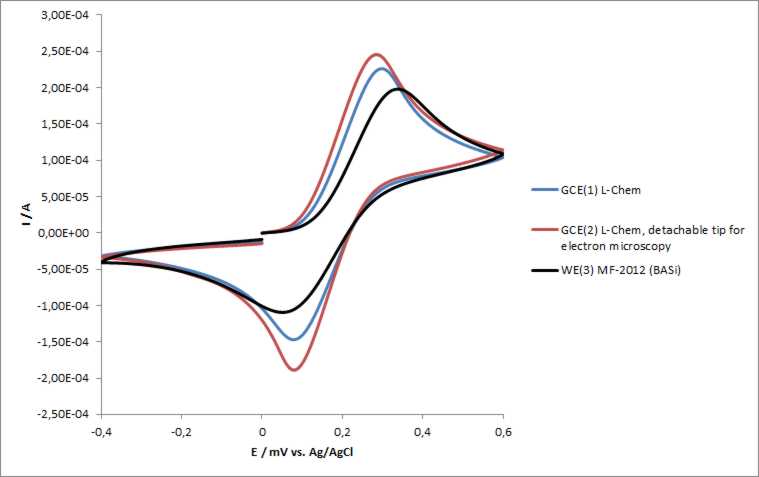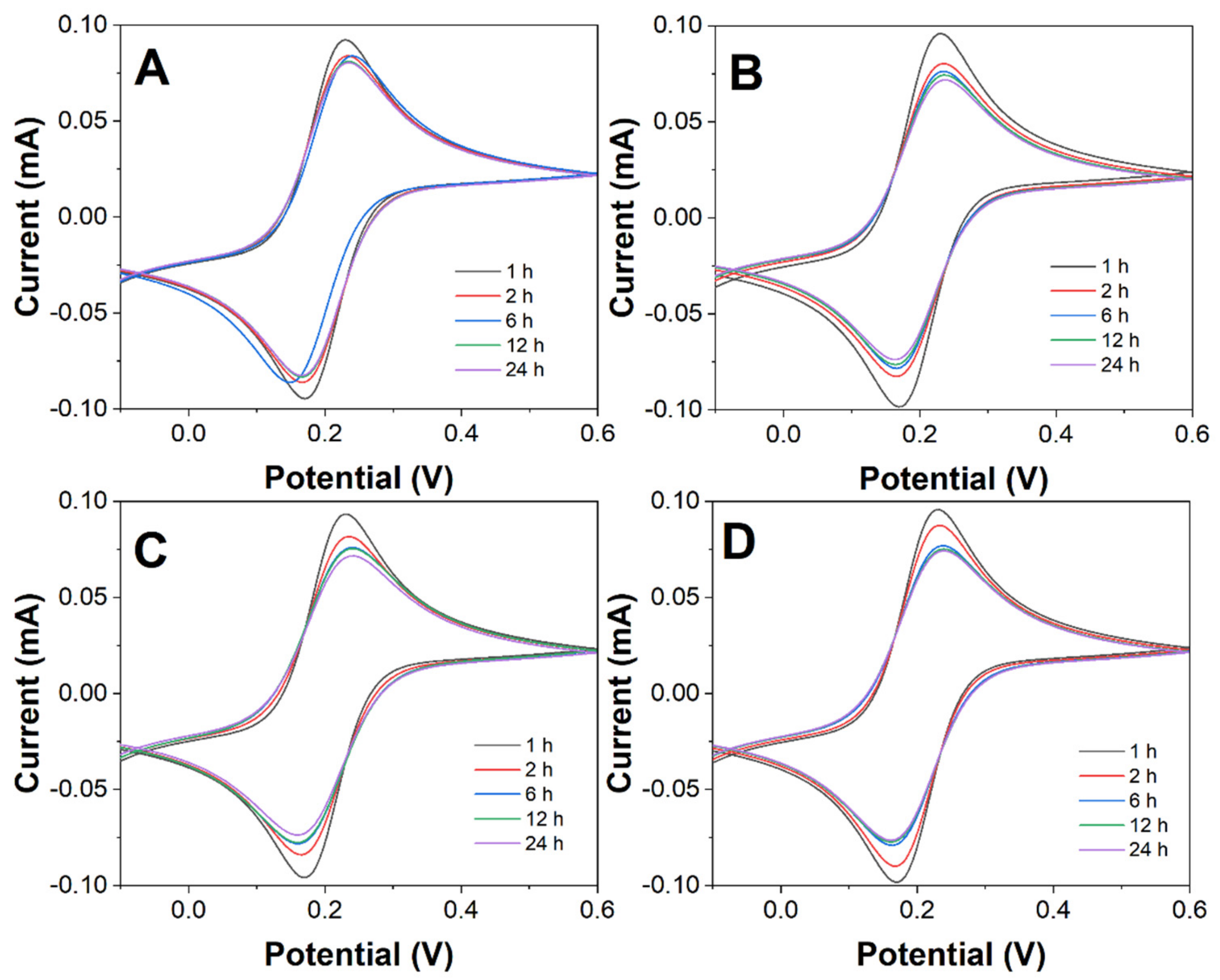
Molecules | Free Full-Text | Electrochemical Immunoassay for Tumor Marker CA19-9 Detection Based on Self-Assembled Monolayer

Oxygenated Edge Plane Sites Slow the Electron Transfer of the Ferro‐/ Ferricyanide Redox Couple at Graphite Electrodes - Ji - 2006 - ChemPhysChem - Wiley Online Library

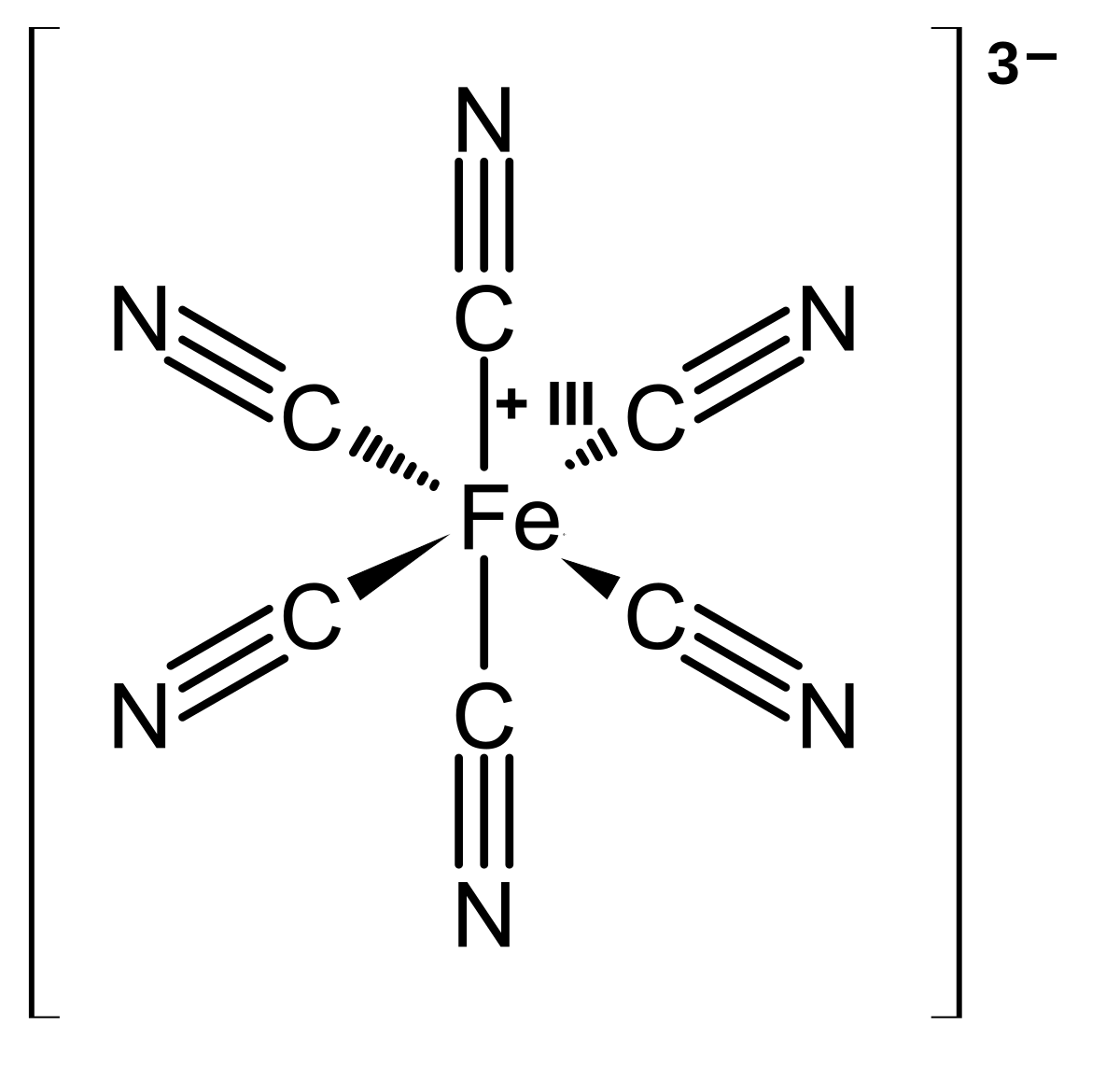
Reversibility of Ferri-/Ferrocyanide Redox during Operando Soft X-ray Spectroscopy | The Journal of Physical Chemistry C

Quartz crystal microbalance electrode modified with thermoresponsive crosslinked and non-crosslinked N-isopropylacrylamide polymers. Response to changes in temperature | SpringerLink

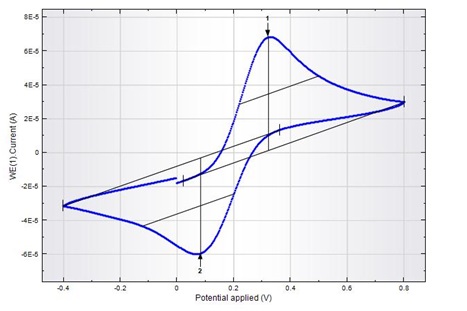
Ferro/ferricyanide couple response studied by cyclic voltammetry and... | Download Scientific Diagram

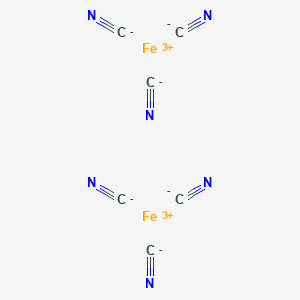
SciELO - Brasil - The preparation and characterization of the hexacyanides immobilized in chitosan The preparation and characterization of the hexacyanides immobilized in chitosan
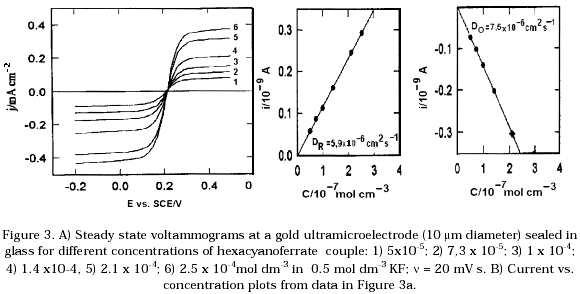
Electrochemical studies of Fe(CN)6(4-)/Fe(CN)6(3-) on gold ultramicroelectrodes varying the concentrations of KF as supporting electrolyte

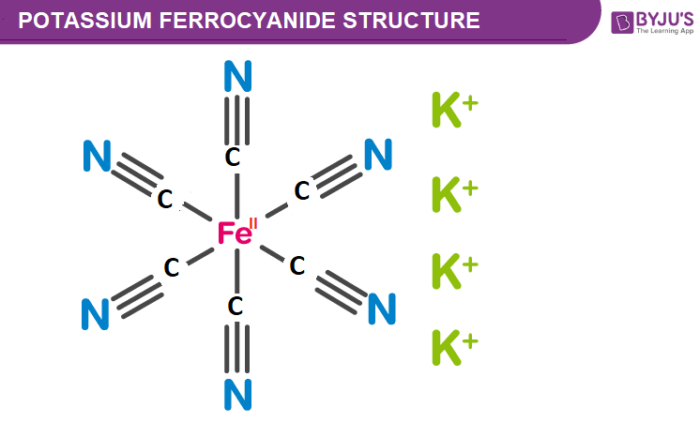
Which of the following statement(s) is (are) correct with reference to the ferrous and ferric ions (a) Fe 3+ gives brown colour with potassium ferricyanide - Sahay Sir

Screen‐Printed Electrodes Modified with Carbon Nanomaterials: A Comparison among Carbon Black, Carbon Nanotubes and Graphene - Cinti - 2015 - Electroanalysis - Wiley Online Library

Electron transfer studies of a conventional redox probe in human sweat and saliva bio-mimicking conditions | Scientific Reports

The study of electrical conductivity and diffusion behavior of water-based and ferro/ferricyanide-electrolyte-based alumina nanofluids - ScienceDirect