Asia-Pacific Roundup: Philippine FDA seeks feedback on abridged procedures for clinical trial applic | RAPS

Press Statement || Updates on Applications for Conduct of Clinical Trials and Emergency Use Authorization of COVID-19 Vaccines - Food and Drug Administration

Department of Health on Twitter: "#ChecktheFAQs! Q: Is there a cure for COVID-19? Why are we vaccinating for this disease? A: There is currently no licensed medication to cure COVID-19. However, there
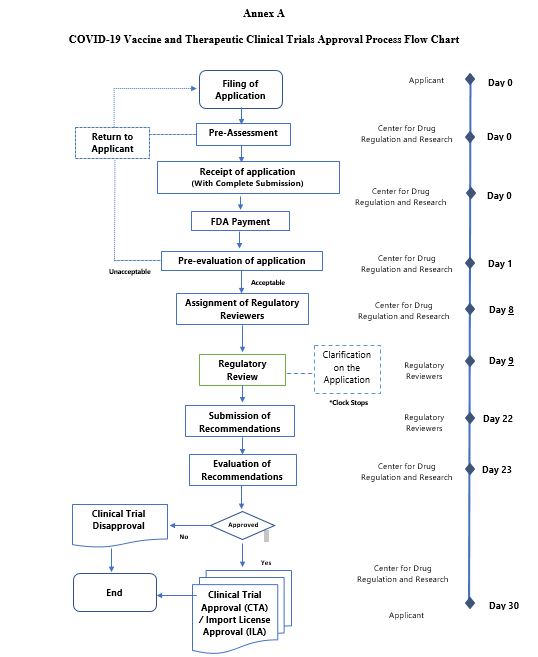
FDA Circular No.2020-029-A || Amendment to FDA Circular No. 2020-029 entitled “Guidance on Applications for the Conduct of COVID-19 Clinical Trials” - Food and Drug Administration

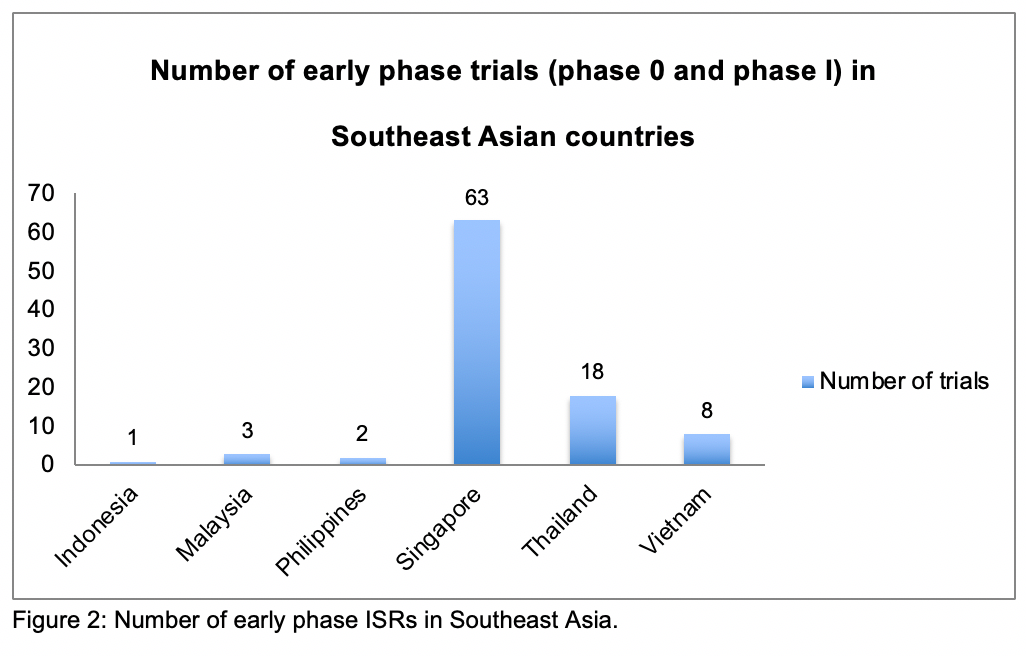
Draft for Comments || Guidelines on Regulatory Reliance on the Conduct of Clinical Trials in the Philippines - Food and Drug Administration

Administrative Order No. 2020-0010 || Regulations on the Conduct of Clinical Trials for Investigational Products - Food and Drug Administration