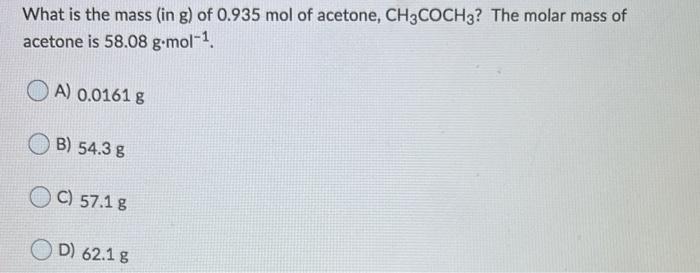SOLVED:1. A student determines the molar mass of acetone; CH;COCHz, by the method used in this experiment The student found that frcezing temperature of water was 1.0 % When the student added

SOLVED: A chemist wants to determine the molar mass of a naturally occurring substance isolated from spearmint. She adds 4.531 g of this substance to 25.00 g of acetone (C3H6O; molar mass
![Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube](https://i.ytimg.com/vi/CCWl-z3Wdmo/sddefault.jpg)
Calculate molar mass of following compounds.i) [CO(NH2)2]ii) [CH3COCH3]iii) [H3BO3]iv) [H2SO4] - YouTube

Acetone Molecule Structure & Chemical Formula | What is Acetone? - Video & Lesson Transcript | Study.com

Sample of mixture of A(g), B(g) and C(g) under equilibrium has a molecular weight (observed) is 80.The equilibrium is A(g) B(g) + C(g) (mol. wt. = 100) (mol. wt. = 60) (mol.


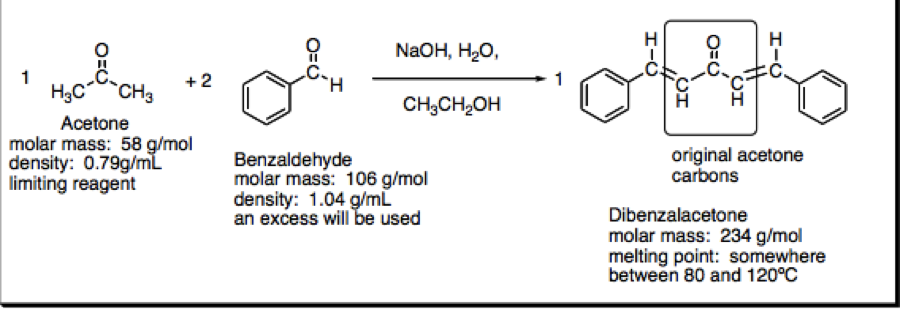


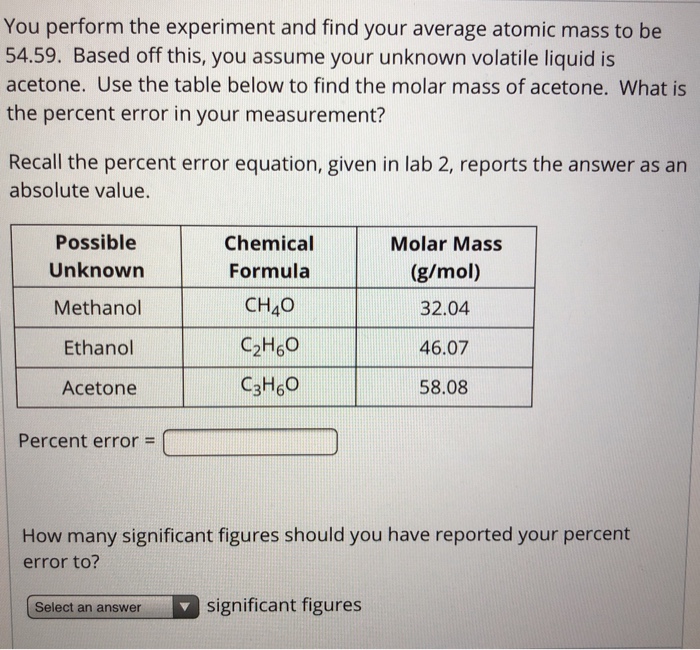
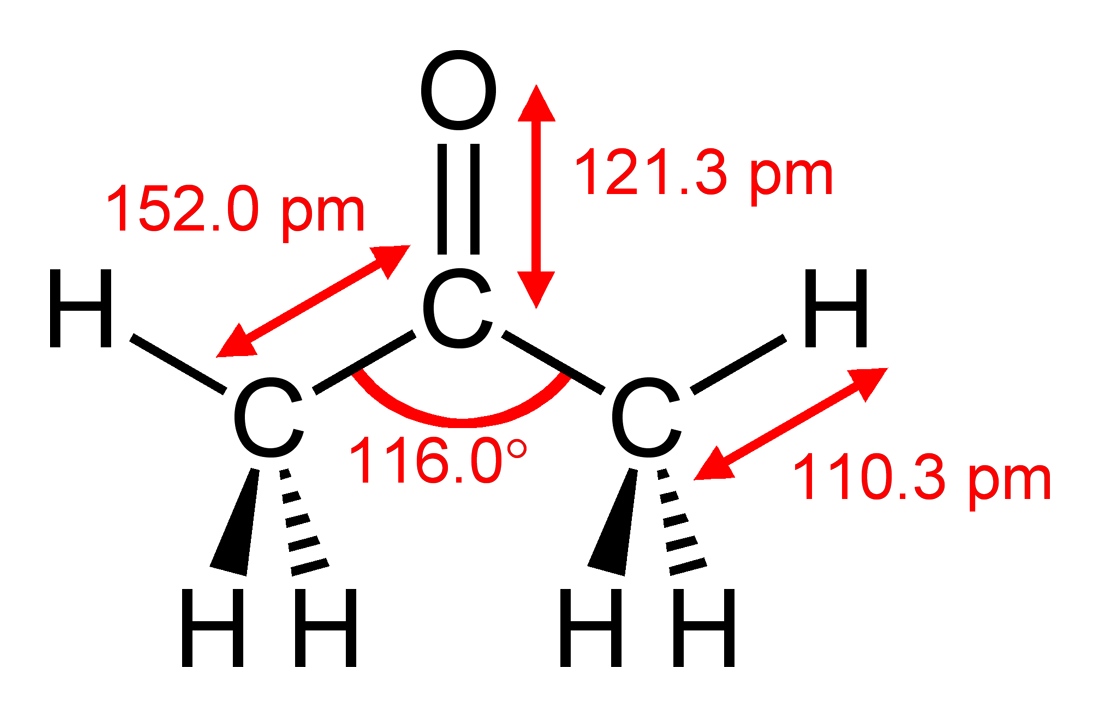


![Calculate the molar mass of the following compounds. (ii) Acetone [CH3COCH3] Calculate the molar mass of the following compounds. (ii) Acetone [CH3COCH3]](https://doubtnut-static.s.llnwi.net/static/web-thumb/378416412_web.png)


![Calculate molar mass [OPENSTAX] Problem 13 - YouTube Calculate molar mass [OPENSTAX] Problem 13 - YouTube](https://i.ytimg.com/vi/DfcoUL7eDL8/maxresdefault.jpg)